ROP continues to be a significant cause of visual morbidity worldwide. Because of improved neonatal care in developing countries, many preterm infants survive [16].Intravitreal anti-VEGF therapy replaces the standard laser photocoagulation for type 1 ROP. For its prompt effect, anti-VEGF can serve as monotherapy for ROP or bridge therapy until more stable conditions are assured for laser treatment. It is also more accessible and easier to administer under topical anaesthesia when compared to laser photocoagulation. However, it is crucial to be aware of the vascular changes and the behavior of treated eyes. Examination of children with indirect ophthalmoscope at pre-school age can be challenging in the clinic. Also, peripheral pathology, vascular remodeling, the boundary between the vascular and avascular retina might be difficult to appreciate using only an indirect ophthalmoscope. FA is a sensitive tool to detect vascular abnormalities in ROP compared with indirect ophthalmoscopy. With FA, we can delineate peripheral avascular area, neovascularization and leakage, and subtle capillary changes [17,18,19].The use of additional modalities to describe abnormalities in eyes with ROP, such as FA and optical coherence tomography (OCT), will probably affect the traditional classification of ROP and reflect on the management and outcomes.
In this study, ten eyes (22.7%) required repeated treatment for disease reactivation; (six eyes; 75% of A-ROP, and four eyes; 11.8% with original zone II stage 3 with plus disease). Second injections were given after 8.6 ± 1.5 weeks from the first treatment. In 2018, Tong et al. studied 160 eyes with aggressive posterior ROP (AP-ROP) treated with IVR monotherapy and reported re-treatment in 82 of 160 (51%) eyes after a mean interval of 7.5 ± 6.9 weeks from the first injection [20].Wong et al. found that reactivation after ranibizumab occurred between the 41stand 42ndweeks [12].In the present study, recurrence was observed in PMA 42.2 weeks on average. Also, a group from Turkey compared the rate and interval of disease reactivation between bevacizumab and ranibizumab treatment for type 1 ROP and found a significant difference in recurrence rate between ranibizumab (50%) and bevacizumab (5.5%). Also, reactivation after ranibizumab occurred at 8.75 ± 1.5 weeks and 14 ± 2.65 weeks with bevacizumab [21].Hoerster et al. measured serum VEGF levels in an infant treated with ranibizumab and showed that intravitreal ranibizumab reduced systemic VEGF in infants for 2–3 weeks, with a return to normal levels at 4 weeks post-injection [22].However, intravitreal bevacizumab showed a reduction in serum VEGF levels for 2 months after injection intravitreally [23].This highlights the importance of studying the risks and benefits of running ranibizumab for its shorter duration of systemic VEGF level suppression and the need for frequent follow-up visits for potential recurrence and re-treatment after IVR.
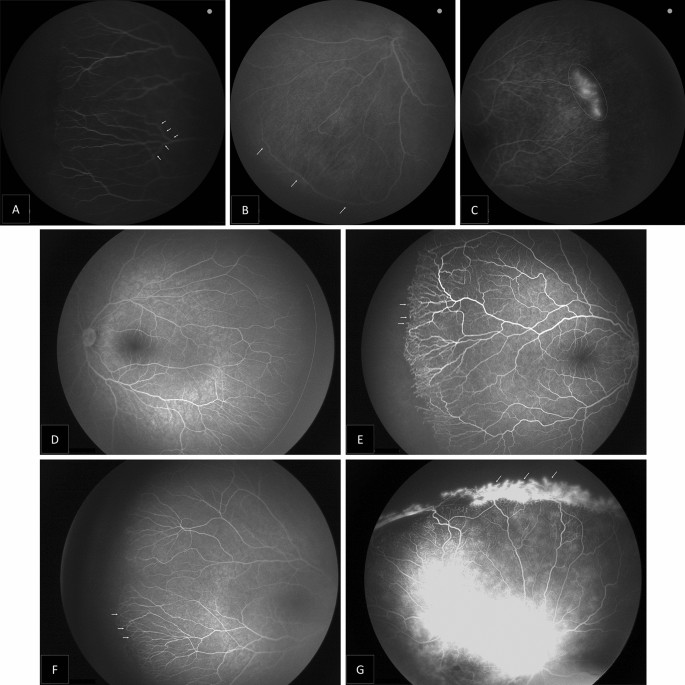
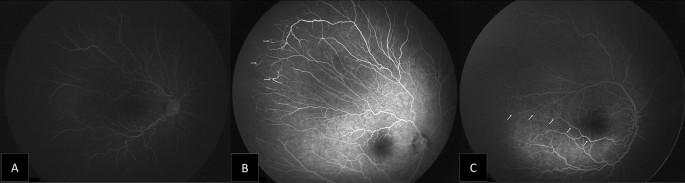
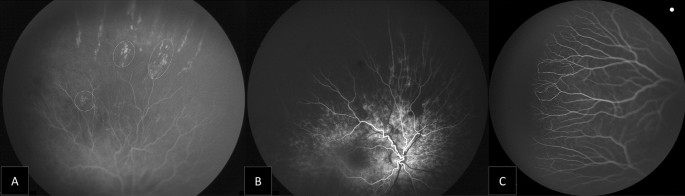
During our FA study, leakage was present in five eyes (62.6%) of A-ROP patients, compared to (5.9%) in eyes with original zone II stage 3 with plus disease. Also, neovascularization was evident only in five eyes (62.6%), all with an original A-ROP who received multiple injections. Mintz-Hittner et al. reported main risk factors for recurrence: low birth weight, low gestational week, and patients with disease in the zone I and diagnosed with AP-ROP [24].Also, ROP recurrence was significantly correlated with more aggressive forms of ROP at initial treatment [25].
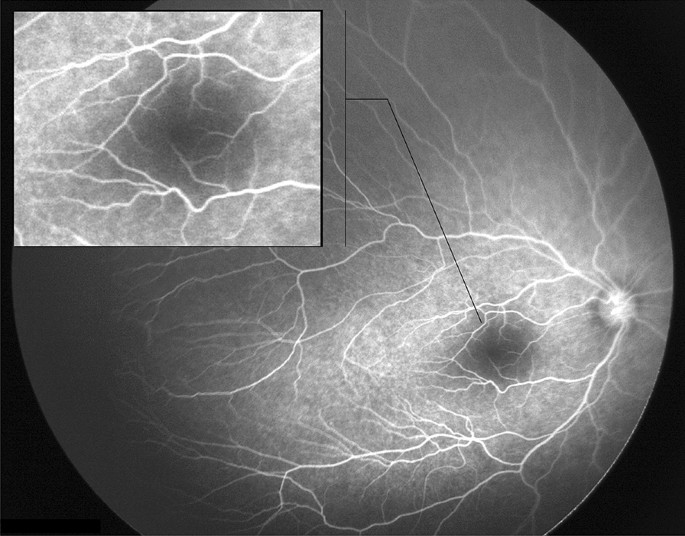
The extent of the vascularity into the peripheral retina is an important aspect of ROP treatment. The avascular retina normally extends ≤ 1.5 disk diameters temporally and ≤ 1.0 disk diameters nasally from the ora serrata in children up to age 13 years [2].However, the persistent avascular retina has been reported after anti-VEGF treatment [26,27].In our study, all 44 eyes did not demonstrate full vascularity of less than two DDs. Also, eyes with original A-ROP and who required a second injection (six eyes) showed extensive peripheral avascular retina reaching Zone I. A retrospective study on 201 ROP-eyes treated with ranibizumab listed low BW, the severity of ROP, and repeated injections as main factors to increase the risk of the retinal peripheral avascular area [15].
Adding to the risk of an avascular retina, ROP eyes have an increased lifetime risk of lattice degeneration, retinal tears, and detachments [28].To date, there are no clear guidelines or consensus on the management of persistent retinal avascularity after anti-VEGF treatment for ROP and no clinical trials on the efficacy and safety of laser in this context. Some elected to apply prophylactic laser for persistent avascular retina in ROP eyes post intravitreal injection of anti-VEGF agents [29].We recommend laser ablation of the ischaemic peripheral retina in the eyes, which showed persistent neovascularization at the vascular and avascular retina junction on FA. A comprehensive discussion on the pathology and possible treatment options with the family is essential. Potential options include prophylactic laser photocoagulation to areas of retinal nonperfusion. Another option is long and close observation with FA study to delay laser ablation for worsening leakage.
The small sample size limited this study’s conclusions. The data collected during patients’ stay in NICU are retrospective. The sample size was insufficient to comment on disparities between disease categories; all patients with type 1 ROP, including those with A-ROP, had similar FA changes. Age at the time of FA was variable among study participants, which allows us to conclude that vascular abnormalities can occur at any age after ranibizumab treatment, but data is not sufficient to comment on age-specific vascular abnormalities. Prospective studies with larger sample sizes must set treatment paradigms and monitoring protocols.